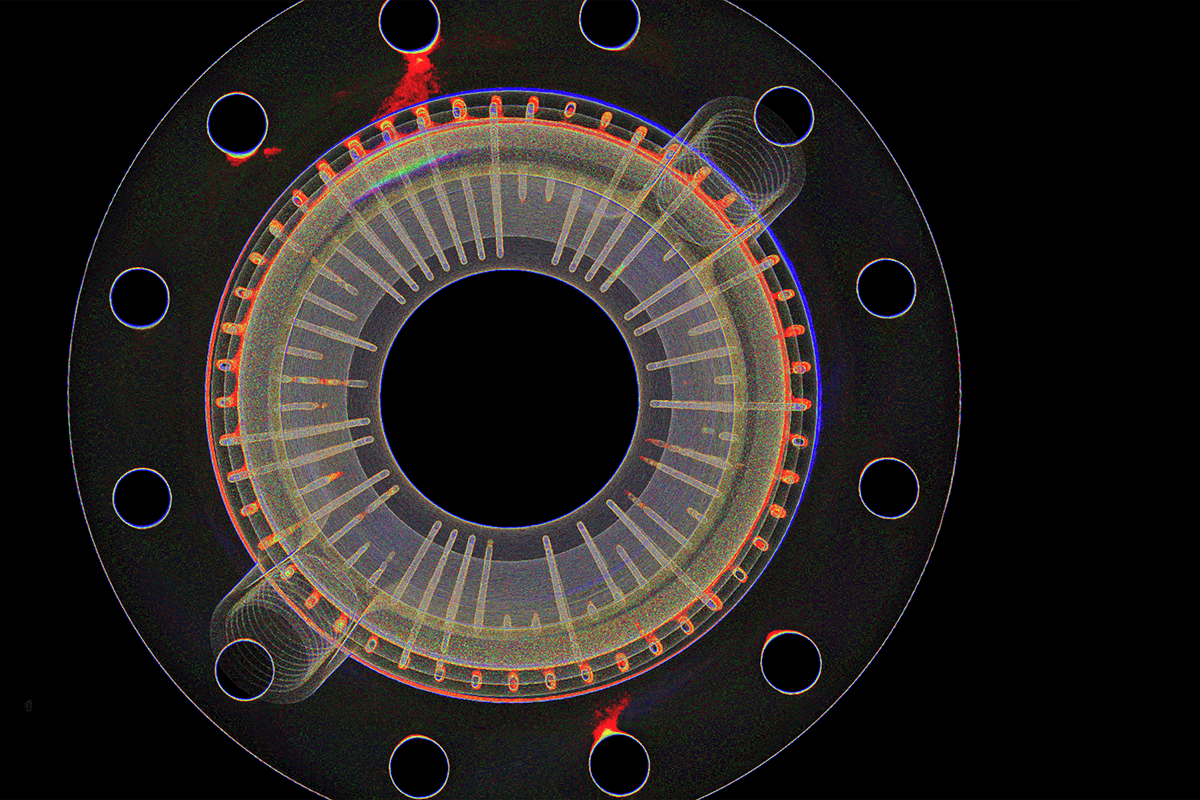
Insulin pump failures are rarely visible from the outside. The Medtronic 630G/670G recall that affected 322,005 devices traced to a broken retainer ring. The Tandem t:slim X2 correction traced to faulty speaker wiring. The Insulet Omnipod field notice traced to a cannula deployment mechanism defect. In each case, the failure was internal, and conventional inspection missed it. This white paper shows how industrial CT scanning gives quality and regulatory teams non-destructive visibility into the components, assemblies, and fluid paths that determine whether an insulin pump works when a patient needs it.
Download this white paper and learn how to:
- Detect fluid path failures including gaps, cracks, failed O-ring seals, and blocked vent membranes that cause silent under- or over-delivery of insulin without triggering an alarm
- Inspect PCB assemblies for solder voids beneath BGA and QFN packages, faulty wire bonds, and battery contact damage from physical impact before these defects cause field failures
- Verify retainer rings, actuators, and drive train components non-destructively, catching sub-millimeter mechanical failures that escape functional testing
- Identify porosity and crack initiation sites in injection-molded housings that compromise IP ratings and create latent failure risk during daily wear
- Confirm cannula straightness, insertion angle, and needle retraction in patch pump assemblies, verifying the deployment mechanism without destroying the device
- Map dimensional deviations against CAD across the full part geometry to catch tooling drift, supplier variation, and out-of-tolerance features before they become a production problem
- Build an audit-ready inspection record, archived, annotated, and reproducible, that supports 510(k) and PMA submissions and eliminates inter-inspector variability in regulated environments
Download the full paper to see how CT inspection covers every phase of the insulin pump lifecycle, from design validation through field failure analysis, with the same non-destructive evidence base.