Medical Devices: Maximizing Inspection Impact with CT Imaging and Analytics

Medical Devices: Maximizing Inspection Impact with CT Imaging and Analytics
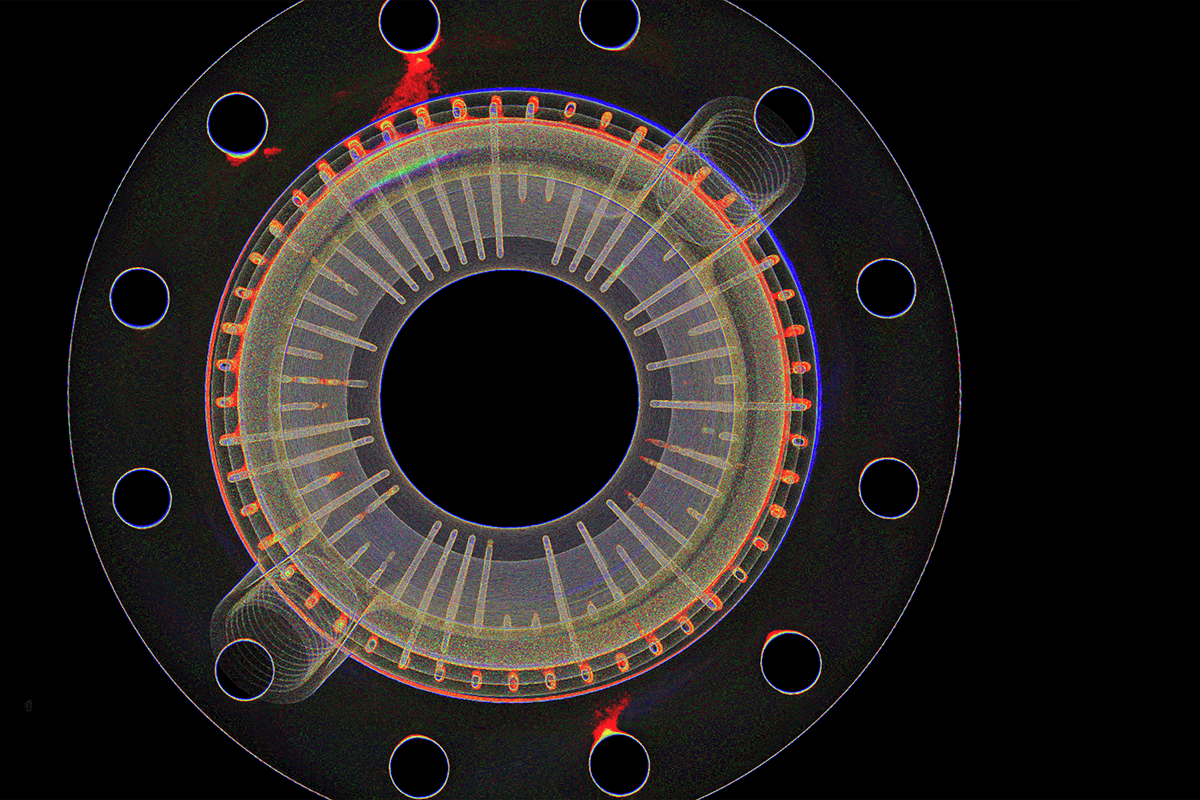
In medical device manufacturing, a defect that reaches a patient is not a quality metric. It is a patient safety event. Yet destructive testing, spot checks, and manual inspection remain common practice, and they share a common limitation: they cannot see inside a finished assembly without compromising it.
This webinar covers how industrial CT scanning makes internal inspection practical across the device lifecycle, from early design validation through production. You will see how CT helps teams find defects earlier, investigate failures without destroying parts, and accelerate verification timelines without adding risk to the manufacturing line. We will also introduce Lumafield's product roadmap and show how integrated CT hardware and analytics strengthen engineering and quality workflows at every stage.
We will cover how teams measure ROI from a CT inspection program, including faster failure investigations, reduced scrap and rework, lower reliance on destructive testing, shorter validation cycles, and higher inspection throughput, all while maintaining the compliance posture the industry requires.
What you will learn:
- How industrial CT reveals internal defects that surface inspection and destructive testing miss
- How CT supports design validation, failure analysis, and production inspection under one platform
- How analytics turn CT data into clear, actionable decisions without slowing the line
- How to quantify the cost and compliance impact of bringing CT inspection in-house
Speakers
Thomas Royston, Head of Application Engineering, Lumafield
Brian McGowan, Enterprise Account Executive, Lumafield