In this episode:
Matt Angle is the CEO of Paradromics, a neurotechnology company building high data rate implantable brain-computer interfaces for people who have lost the ability to speak and move. The company's Connexus system, currently entering clinical trials, clocks an information transfer rate of over 200 bits per second (more than 20x the reported performance of comparable systems) and is designed to last more than a decade in the body.
The conversation covers the basic physics of what separates an implanted electrode array from a surface EEG, the design of the Connexus itself, and the engineering choices that distinguish Paradromics from Neuralink: hermetic sealing, modular battery placement, and a deliberate bet on durability over planned obsolescence. We also get into what it takes to manufacture a device at the micro scale, where CT scanning fits into quality control once you've welded a titanium case shut, and the formative go/no-go moment when the team bet their company on a DARPA contract against consortium bids from Berkeley, Harvard, Stanford, and UCSF.
Links from the discussion:
Paradromics: https://www.paradromics.com
Connexus BCI for people unable to communicate: https://www.paradromics.com/product
SONIC benchmarking standard: https://www.paradromics.com/blog/bci-benchmarking
SONIC preprint (bioRxiv): https://www.biorxiv.org/content/10.1101/2025.09.30.679683v1
Tempo BCI for mental health brain-state monitoring: https://www.paradromics.com/tempo
Transcript:
Welcome to Go/No-Go. I'm Jon Bruner. This is a podcast about design, engineering, manufacturing, and the calls that can make or break great products. Go/No-Go is brought to you by Lumafield, which makes a manufacturing intelligence platform that uses X-ray CT technology and artificial intelligence to give engineers total confidence in the products that they're shipping. My guest today is Matt Angle. He's the CEO of Paradromics, a startup making a novel brain-computer interface. Matt, it's great to have you on.
Thanks a lot for having me.
What is a brain-computer interface?
Before I explain exactly what a brain-computer interface is, I'll start by telling you a little bit about our brains and how we experience the world. Everything that we hear comes to our brain as neural signals from our ear. Everything that we see comes as neural signals from our eye. Everything that we feel comes as neural signals from our sense of touch. And everything that we do originates in the brain and goes to the muscles as neural signals. Neural signals are the currency of human experience. So when you ask what a brain-computer interface does — it reads and writes neural signals. In some sense, there's very little that a brain-computer interface can't do. But if we talk about near-term applications and what we're using it for right now, it's for restoration of function. We're currently in a clinical trial building brain-computer interfaces for people who have lost the ability to speak and are paralyzed. For someone who is paralyzed and can't speak, a brain-computer interface can be implanted in the area of their motor cortex that would have told the muscles forming speech how to speak. Instead, we can infer their intent and create speech through a computer.
So these are disabilities where the brain has lost its connection to some other part of the body, and you're able to recover some of those signals and send them to a computer.
Exactly. And the same kind of device, placed in a slightly different area — the area that would control the arm — can give someone who has lost their arm the ability to control a robotic arm. Move it to yet a different area and it can actually read out brain states related to things like attention and depression, which offers a really interesting way to reframe conditions in mental health. It's a highly versatile platform.
And in those mental health cases, you'd be writing to the brain — modifying its function or its understanding of the world.
For sure. And in some sense, for mental health applications, you don't even have to write to the brain. Sometimes just reading can be interesting. I give the example of a continuous glucose monitor — all it does is read your glucose levels, and yet it's transformed the treatment of diabetes because just providing data can help you adjust the existing mechanisms for managing that disease.
There are some non-invasive ways to read brain activity, like EEGs. How does an implant give you something that an EEG doesn't?
Neural activity is at the base of existence. The substrate for that activity is individual neurons — little cells inside your brain. You have 85 billion of them, and they coordinate their activity to encode information. The basic unit of information in the brain is a record of whether neurons are firing, called action potentials — quick, binary signals they use to communicate with each other. If you have a device inside the brain with electrodes that reach down into the layers of brain tissue and get close to the neurons, you can record individual neuron signals and know at the highest granularity what's going on. If you're sitting on the outside of the skull, you're listening to the correlated activity of millions of neurons. You can only know when something's happening when a lot of neurons are coordinated and doing the same thing. The physics analogy people often use is: imagine dropping microphones into a stadium of 100,000 people versus having microphones in the parking lot. In one case you can record the conversations; in the other, you might know if someone scored because everyone shouts at the same time.
Much finer grain detail and higher bandwidth.
Exactly. The key to bandwidth in a brain-computer interface is whether you have the resolution to resolve individual neurons, and how many neurons you're talking to.
How many neurons are we talking about with the Paradromics system?
Our first system is a cortical module that sits on the surface of the brain and has 421 electrodes, so it can talk to roughly 421 neurons. We built the system to be modular so that eventually we can implant multiple cortical modules to access different areas. Going forward, I see us roughly doubling the number of electrodes per unit area every three years — a kind of Moore's Law of brain access.
Describe what the implant looks like.
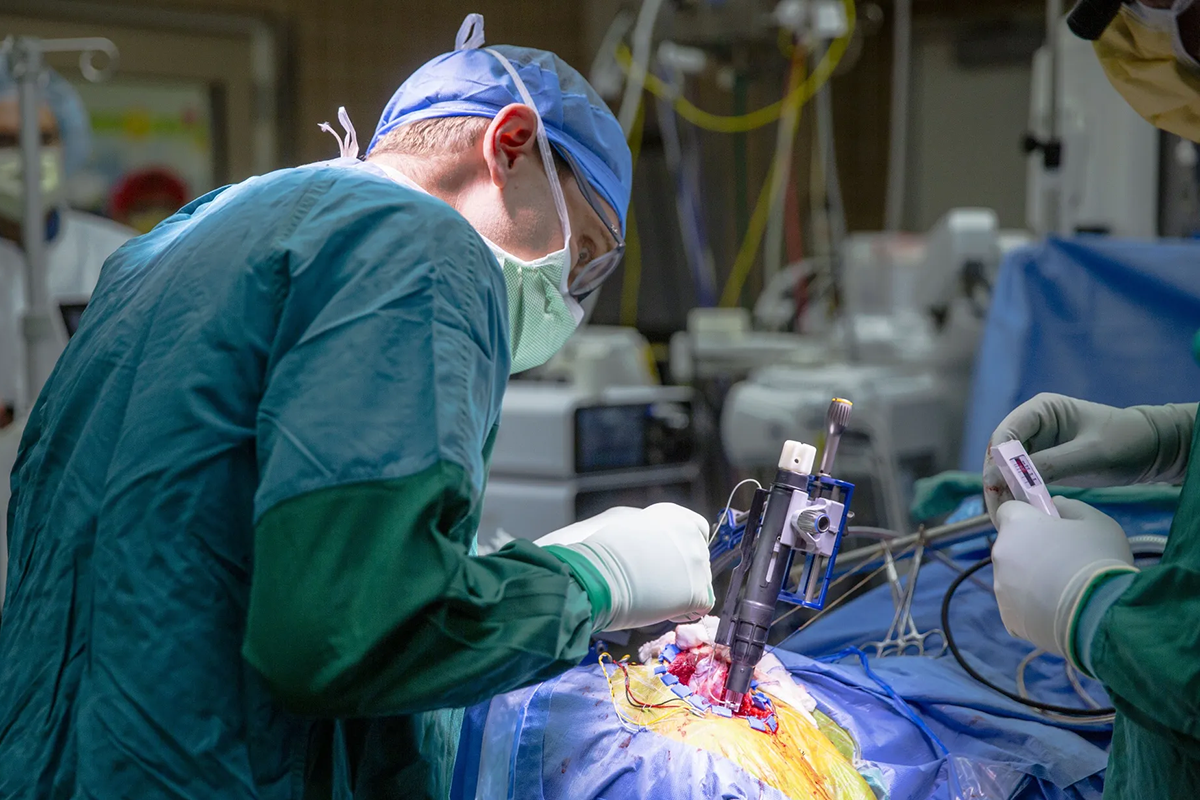
It's smaller than a dime and has tiny platinum-iridium micro wires coming off of it. If I were smart I'd have one in my hand today — we'll send a picture to the producers. 421 little wires, each smaller than a human hair. They're 40 microns in diameter and 1.5 millimeters long. They insert down into the cortical tissue at the output layer of motor cortex, where the neurons most active in controlling the body are. They're sitting in just the right area to understand what someone is trying to do when they attempt a movement. That signal is transmitted through a lead that runs under the skin — the whole system is hidden. Then there's a wireless power and data unit that sits in the chest, charged wirelessly, that transmits data wirelessly.
So there's a battery in the chest along with the ICs involved in transmitting the data.
In this case the battery actually sits on the wheelchair and the device is continuously inductively powered.
Like broadcasting energy into your chest the whole time.
Exactly. It's like how you charge an electric toothbrush.
Do you see this becoming a consumer product or is it only a medical therapy on any reasonable timeline?
Our short-term goal is to use this technology to help people by building restorative therapies. But the capabilities we'll be building for people with disabilities are in some sense dual use. The first people who get speech prostheses are going to have the ability to query large language models directly from their brain. They'll start interacting with AI in a way that's slightly different from how you and I do — and you could already consider that a superpower. As BCI expands into, let's say, a hearing BCI, a trivial application to run on top of it — depending on how much latency you're willing to accept — is automated translation. Someone who gets their hearing through a BCI may suddenly be able to understand a hundred languages. What you'll have is this accumulation of really interesting abilities in the disability community and a record of phenomenal safety. And then you could have a situation where people with no disability at all look at that and say, I'd be interested in that.
Skipping over the era of the Humane Pin or the earbuds in your ear doing translation — you would be experiencing these things directly in your brain.
In some sense it may be very similar to the experience you or I would have with earbuds or a Humane Pin, but in other cases it may actually short-circuit some of the ways we interact with technology. As BCIs get more sophisticated and start recording from areas upstream of direct motor output or downstream of direct sensory input — particularly as devices begin working in areas related to attention or cognition, which they will naturally do as part of the quest for understanding mental illness — we'll start to develop a deeper awareness of what's going on in the brain and see opportunities to tie into technology in ways that maybe we wouldn't even think of today.
Let's talk about where the product is right now. You've been testing in sheep for several years.
Exactly. We have a cortical module we've been testing in sheep. The system, called the Connexus, is the highest data rate system in the world — we clocked it at over 200 bits per second in our sheep model. We now have FDA approval to use the same device in a clinical trial for people who have lost the ability to speak and have quadriplegia. In the coming weeks, we'll be implanting a patient with quadriplegia with this device and allowing them to communicate again.
How has that FDA process changed the product development workflow? Medical devices often take a really long time to bring to market.
It's helpful that we've been able to benefit from FDA programs like the Breakthrough Device program and the TAP program. Those have been good for closing the loop with regulators quickly and understanding where they are. But there's still a fundamental challenge to moving quickly and designing iteratively in the medical device field. In an unregulated field where the stakes are relatively low, the fastest thing is to test systems separately, integrate them, get a proof of concept, swap out parts — you're constantly improving and making judgment calls about what could be swapped in without breaking the whole system. In a regulated environment, particularly with the FDA, they want to see you test an entire system and then have that exact system go into the clinical trial. There's a really detailed change management process if you want to change anything. It works against the quick-moving intuition you might otherwise have as a technology developer.
What did the very first prototyping stages look like? Were you iterating in animal models, or are there other ways to model brain activity in a lab?
There aren't any very good ex vivo models for neural recording. In the end you really do have to do an end-to-end system test, and for brain-computer interfaces it has to be in a large animal. But one of the things we knew coming in — because the iteration cycle is so slow and we didn't want to take too long or spend an absurd amount of money — is that we wanted to lock down a lot of aspects like materials and core architecture decisions, and stay within the lines of what had been successfully demonstrated previously in human studies. There had been a number of clinical trials preceding us using older BCI technology. We knew what worked and what didn't. We knew you could transform signals from motor cortex into cursor control, robotic prosthetic control, or imagined speech. So we knew that if we could recapitulate those experiments with a device that was substantially similar and recorded the same types of signals, our trials would be successful. We engineered the areas where on first principles you could clearly see that changes wouldn't add additional risk. But we stayed close to precedent in places like materials, biocompatibility, and hermetic encapsulation.
Would you say the fundamentals of BCI are pretty well understood and this is now in a Moore's Law stage of technological development — just packing more sensors into the devices? Or are you discovering new things about the fundamentals of brain activity all the time?
The fundamentals of recording brain signals, decoding brain signals, how neurons and populations of neurons encode information, and how to build a BCI system — those are very well understood. There are application-specific questions for any given disease or placement in a certain brain area that are often situation-specific. That's actually one reason we recently created what we call our APEX program — our application expansion program — where we partner with academic labs around the world that have expertise in recordings in certain brain areas, provide them with our hardware, and run clinical trials together to better understand new indications. When the results are positive, we then launch full clinical trials in those areas and bring products to market. There's still a last mile on the application front that leaves room for discovery, and industry-academic collaborations are essential there.
I imagine a real brain-computer interface in someone's brain has been a pretty rare experimental resource up until now. You're foreseeing a world where a lot more people have these, and it'll become possible to better understand applications pretty rapidly.
It's definitely a flywheel. We've been limited by the availability of human-ready hardware. Now that we have a platform that is not only ready for robust clinical research but also ready to go to market as a product — something that eventually thousands, hundreds of thousands of people will get — we'll start generating a lot of data from patient populations that can in turn improve outcomes not only for the population it was initially intended for, but also to discover biomarkers and suggest therapies for totally different indications.
That sounds like an extraordinarily difficult device to manufacture, especially at the one-off prototype stage you're in now. What does manufacturing actually look like?
There are a number of steps where we can rely on traditional medical device manufacturers, and then a few steps where it's quite specialized — everywhere we're trying to do things at the micro scale. The microelectrode formation, bonding them onto what's called a feedthrough to keep the whole thing hermetic so moisture doesn't get inside, the microelectronic assembly — those are things we specialize in. Everything else — lead production, traditional implantable casing — are places where we work with partners who have a good track record building those kinds of things.
How do you think about quality and life cycle for these devices?
Especially this early in the process, when we're developing the manufacturing process alongside the design and it's still small artisanal batches — quality along the way and checking many, many times throughout the process, really understanding where failures occur and being able to track them, is essential. Even after devices are sterile packed, we have additional quality steps that occur in the OR before a device is ever implanted. In these really early stages we just want data at every stage, and we want to be absolutely sure that no patient would ever see a non-functional device. Quality is essential in medical devices, and it's almost a competitive advantage for early-stage companies that do it right.
What are the failures that are hardest to catch? What worries you?
In any manufacturing process it's always the stuff at the very end that's scariest. We never want to be in a situation where the neurosurgeon opens something up, we test the device on the bench, and it doesn't work. We have other inventory, so we'd grab something else. But the later a failure occurs, the more valuable what you're losing, and the closer to the patient it is. There are certain steps where yield loss is just accepted because the raw materials are inexpensive. Instead of spending an absurd amount of money fixing that one step to be really high yield, you accept that you do an inspection procedure and toss the bad ones. That's not good if you're close to the patient. The closer to implant time, the more serious any failure becomes.
Is this a pretty standard surgery? Could any neurosurgeon do it, or is it a specialized form of interacting with the brain?
The device is highly specialized but the surgery itself is pretty standard. Any credentialed neurosurgeon can do a craniotomy — it's table stakes in the field. It involves opening a window in the skull and exposing the brain surface. What the surgery looks like is: expose the brain surface, bring in our device, which is an inserter that looks a lot like an EpiPen. It has the electrode array at the end, and when it's close to the brain surface, it taps the electrodes into the brain and then withdraws. Then we have a lead that comes out of the skull and is tunneled down under the skin — the same procedure used for a deep brain stimulator or a shunt for intracranial pressure. A lot of neurosurgeons know how to do that, and it's not an adoption barrier. These are quite straightforward.
What technological advances are you looking at that'll move this forward in the next few years in terms of manufacturing processes, materials, or processing power?
We're very lucky in that a lot of the things downstream of the BCI — edge computing, AI — are moving way faster than BCI. In terms of computation or models, I don't think we'll ever be lacking for those. The critical bottleneck, in my opinion, is around data. One of the challenges for building high data rate systems is to build systems with a lot of electrodes at a very fine scale so that you can interact with the brain without disrupting it and get a lot of information — but also have those things last for more than a decade in the body. There are probably two dozen labs in the world that can build high channel count recording systems that will last for 30 days. There are probably two dozen companies that build robust implantable neurological devices. But the intersection between building things that will still be there 10 years from now and high channel count recording capabilities — that's just one company, and that's us. Anything that enables you to get more data in and out of the brain without sacrificing durability — those are the kinds of material and packaging breakthroughs that will be exciting.
You mentioned other labs working on BCIs — Neuralink famously, and a couple others. Can you characterize how your approach is different from the three or four companies people might have heard of?
There's a big divide within the BCI community between whether you're optimizing for data rate or something else. A number of companies have come up with novel ways to deliver electrodes to the brain or record signals from outside the brain. They're betting that the markets they're interested in will only be accessible to devices that aren't implanted surgically, and that those markets will be accessible with low data rates. That's not where we play. We're solidly in the high data rate camp, which means implantable. The only other company really in earnest going after that is Neuralink. They're a super smart group of people, very well funded, and like him or hate him, Elon's one of the best tech CEOs in the world, possibly in our lifetime. They're formidable. But we don't agree on starting points for BCI. We looked at the space and immediately said the next 20 years of people getting implantable BCIs are going to want a commitment that these devices will last more than 10 years. If you talk to people who are BCI users now or people considering brain implants, everyone is nervous to get brain surgery even once, and no one wants a device that's going to fall apart in two years. That was an important design input for us: build something that lasts a long time. Elon is designing for a 50-year future where everyone is comfortable getting brain implants implanted and removed — a planned obsolescence for brain implants, like phones, turned over quickly. An outpatient procedure at a strip mall somewhere. But that's so many decades out from where the world is now that for a company like us, backed with venture capital, our investors don't have that much patience. And frankly, there are so many healthcare opportunities to deliver for people in the near term that I'm happy to build robust devices. The other thing is that you would think theoretically that building a less robust device — something that has to be removed every once in a while — would open the design space and allow for higher data rates or some other advantage. But empirically, the Paradromics device is clocking a higher information transfer rate than the Neuralink device. In practice, we haven't sacrificed anything by making our devices robust.
How do you design a piece of electronics that can last for decades inside someone? What does that electronic design process look like?
The first thing that kills an implantable device is moisture — ingress of water and ions. The first thing you can do is make sure the device is sealed up tight so that the environment it sits in is dry for as long as you want it implanted — which means 20 years or more. That's already the first point of departure between the Paradromics and Neuralink devices. Our device is hermetically sealed so the electronics will be protected from the moisture of the body indefinitely. The Neuralink device isn't hermetic. Fluid washes around inside it — it's not leaking like the Titanic, but it's a slow ingress of water vapor. You can put desiccant in there. It's a time-dependent process. You can time it out and say it's okay — it's not going to last more than two to five years anyway, so who cares? That's a rational decision. It's not one we've made, but it's not an accident either.
Why wouldn't they seal it? Is there an extra cost to sealing it?
Their system is so different from ours. I'll just say that it is more challenging to make something hermetic. It means you have to use materials like metals and ceramics. You can't glue a metal to a ceramic — you have to braze it. There's a whole stack of complexity when you're trying to make something hermetic, which in my view is worth it because then you know the device can last for more than a decade. And I'll say — CT is really cool for this. When you seal something up, you have this titanium case that gets welded shut, and if there's a solder contact missing inside, you turn it on and it doesn't work and everyone goes, oh no. But with CT, the resolution is so good that you can see exactly where the soldering went wrong. There's a lot that can go wrong after you close something up, and CT is perfect for that — in situ inspection. It's an awesome tool.
You mentioned batteries as another time-limiting factor for implantable electronics.
Our first system was for wheelchair users, so we didn't even need a battery — they carry one with them, and we chose to continuously inductively power the device. Future devices will be for people who are ambulatory and will have a battery. But even then, thinking precisely about where you put the battery matters. In our device, the battery is implanted in the chest. The barrier for swapping out a chest unit is quite low and simple — you don't have to touch anything in the brain. But if you had a device anchored in the skull and irretrievably connected to the electrodes in the brain, when the battery dies you have to pull the whole thing out. These modularity decisions are important because inevitably you will run down the battery — whether it lasts 5, 10, or 15 years. You want a way of separating that from the brain component.
In terms of common implants like pacemakers, how does this life cycle compare to standard design guidelines for those?
The expectation for implantable electronics is to last more than 10 years, and many last more than 20. It depends on the exact device. There are often other failure modes — like in the leads — that can limit a device's lifetime, but the electronics are expected to last a long time.
To return to our earlier conversation — your first-generation product is aimed at helping people with serious neurological disabilities, but there's perhaps a future where this changes the way people without disabilities interact with AI. What's required in terms of the progression of your design and manufacturing to make that possible?
I'll push back a little. I think it can already be a widespread technology even within the disabilities community and the mental health community, because the number of people who could benefit from a brain implant for therapeutic reasons numbers in the millions. The reimbursement you can get for a medical device of that nature can be in the tens or even hundreds of thousands of dollars. Morgan Stanley estimated the biomedical total addressable market for BCI is around $500 billion. Now, to your real question — the barriers to expanding into everyone. I don't think there are important physical or design barriers that would push BCI from therapeutic to an everyday technology. I think it's more practical and societal. Everyday use doesn't get reimbursement, so the cost of the device would have to come down to consumer levels — iPhone levels. That might be a manufacturing challenge for people to tackle down the road. There's also a basic science component to many of the things we imagine when we talk about superhuman capabilities — it just hasn't been borne out in clinical trials yet. No one's going to want to get a device until they know the benefit they're looking for is actually there. And then there's safety. BCIs are very safe already, but there's still some non-zero risk of infection, and there's an unknown aspect in that there just haven't been that many BCI surgeries for us to talk about the statistics empirically. Until at least a thousand people have had BCIs and the safety statistics are very clear, that will be a barrier for people wanting to get one for, say, video game use.
People want to see that it works and that it's safe.
Exactly. But from a fundamental physics, materials, and design standpoint, the devices being made today and the ones on roadmaps for companies like Paradromics and Neuralink are already quite compelling.
I'd love to move on to the final question we ask every guest. Matt, please tell us about a go/no-go moment you've faced. How did you handle it?
Running a BCI startup for 10 years is really a series of sprints and a series of go/no-go moments. But one of the really important ones — and this was a place where I had to make a founder-level decision — was early on, maybe a year to a year and a half into Paradromics. We were building a system partially supported by NIH and partially by angel investors. It was usable for research, could be used for animal experiments where the device is outside the skull, and had a really high data rate — even higher than the implantable system we're using now. We were on a path to bring this to demonstration and sell it to labs as a tool. That wasn't where we wanted to go. Where we wanted to go was building implantable systems for biomedical applications. But it looked like we could get some early traction and early revenue, and for many of our early investors it was an attractive way to de-risk the company. Then I found out about a DARPA program focused on high data rate BCI — Defense Advanced Research Projects Agency — and started to get the sense that we should really be part of this program. I met the program officer. It was a real long shot. The other groups applying for money in the same funding call were consortia of universities like Berkeley, Harvard, Stanford, and UCSF. All the established names had worked with DARPA before. We were a group of probably eight people working out of what was basically a warehouse space. Our neighbors were an organic cracker factory, a daycare, and a marijuana dispensary. We bet essentially everything on winning this contract against some really big names in the field. A lot of people thought we were crazy to divert almost all of our effort as a company to capturing it. We ended up not only being one of the performers who got funded, but in the end I would say the most successful performer in the program and the only one that resulted in a real product at the end. If we hadn't made that leap, we almost certainly would have gone out of business.
You took the moonshot. What do you credit your success to, going up against all the old guard at the universities?
We had a lot of support from some of our academic collaborators, but mainly we just put in more time than they did into our application. We thought through everything. And when we initially got a small amount of money to prove things out, it was existentially important for us. They were treating it like just another grant. We considered it: we have to deliver on this program or we will not have enough money to make payroll six months from now. We took it more seriously. And I also think we just had a more practical technology bet than some of the others. On both fronts — motivation and technology — we were more motivated, more practical, and more outcome-oriented than a research lab would have been.
And that still kind of drives you.
To some extent. We're more resource-limited than some of our competitors, and that drives us to make more practical decisions. But in a design space this open, sometimes having too many resources can be the kiss of death — you end up chasing too many dead ends or committing in a path-dependent way to something that's not a good idea. We constrained ourselves early on and ended up with a really good design. Although I'm certainly glad that in the fullness of time there are many more resources available. BCI is hot and we aren't primarily driven by poverty anymore. The BCI investment scene is pretty exciting now.
How do you stay focused with all those resources?
Practice. You've learned it the hard way. Muscle memory.
Terrific. Well, it's been wonderful speaking with you, Matt. If listeners want to check out what Paradromics is doing, we'll include a link to Paradromics in the show notes, along with a picture of the Connexus system — I'm sure my verbal description was unsatisfying. I've also seen CT scans of the implant that some of your team members have shared with us, and it's a remarkable device. Thank you so much, Matt. It's really been a pleasure.
Thank you so much for having me.
If you'd like to learn more about Paradromics, check out the show notes attached to this episode — we'll include links to their work and images of the brain implant device. And if you'd like to get in touch with us at Go/No-Go, send us an email at gng@lumafield.com. We always love hearing from listeners. If you enjoy the show, please leave us a rating on iTunes or wherever you listen — it helps this podcast find its way to people who might enjoy it. Go/No-Go is brought to you by Lumafield, which uses X-rays and AI to give manufacturers and engineers total confidence in the products they ship. You can learn more at lumafield.com. For Go/No-Go, I'm Jon Bruner.
Go/No-Go is brought to you by Lumafield, which was founded to upgrade manufacturing. You can learn more at lumafield.com. Go/No-Go is produced by Austin Carder and edited by Brian Tran, with additional assistance from Eric Petralia.
Articles Featured in the Episode